The ONELITE serialization system encodes alphanumeric digits and one-dimensional and two-dimensional barcodes such as GS1 DATAMATRIX.
This is complemented by the automatic inspection system, in charge of corroborating the information contained in the code through the quick capture of the 2D code reader and its immediate inclusion in the database for unit dose stock monitoring.
Compact, portable and versatile equipment. It optimizes spaces in medical establishments and prioritizes its easy operation and setup, allowing the processing of medication formats by simply exchanging its central disk.
HTH Healthcare Line: PUL-ONE | For Patient Safety
Automatic single-dose packaging module with a traceability system PUL-ONE
Automatic equipment designed to fractionate and package tablets, ampoules or vials in unit doses of medications. With an integrated software system for serialization according to the traceability requirements of pharmaceutical products.
Details Gallery
Print and check.
Interchangeable disc.
PUL-ONE + PUL-LITE PLUS
Outstanding Features
Necessary technology to improve the safety of high-risk medications.
- Versatile; processing of different medication presentations allowed in the same equipment.
- Total traceability from the pharmacy to the patient.
- Quality according to national and international guidelines.
- Stock control in detailed reports.
- Compact and portable.
- Plug & Play; only electrical power needed. Airless technology.
- Cost efficient; reasonable cost consumables of national origin guaranteed.
- Adjustments allowed by operator-friendly 15” touch panel.
- Product made in Argentina with technical support throughout the national territory.
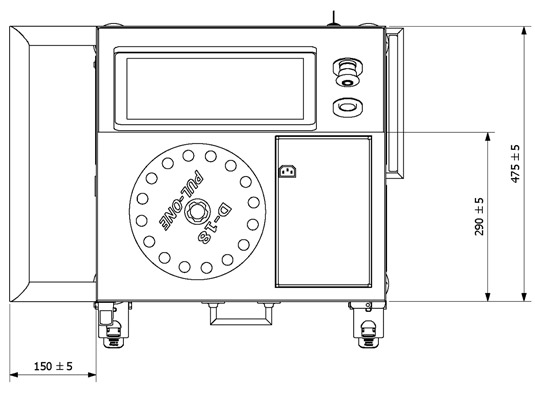
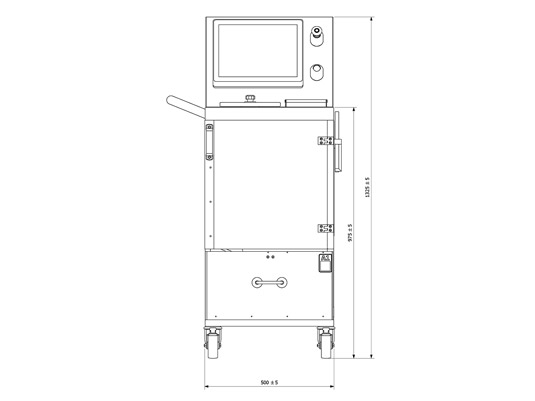
Actual Dimensions
Upper Layout
Front Layout
Technical Data
| Dimensions L x A x H (mm) | 520 x 680 x 1330 |
| Hand-working height (mm) | 1000 |
| Voltage (v) | 220 (50 Hz) |
| Weight (kg) | 100 |
| Installed power (KVA) | 1 |
| Noise level (dB) | <70 |
| Production capacity | 30 unit doses/minute |
| Pre cut blister L x A (mm) | 23 x 45 |
| Ampoules - Specification (ml) | 1 / 2 / 3 / 5 / 10 / 20 |
| Ampoules - Diam x Alto (mm) | 10/22,5 x 35 / 110 |
| Vials - Especificación (ml) | 2R / 4R / 6R / 8R ISO 8362-1 |
| Vials - Diám x Alto (mm) | 10/22,5 x 35 / 70 |
| Admissible plastic wrap width (mm) | 75 |
| Packaging length (mm) | 70 / 160 |
| Printing area A x L (mm) | 30 x 50 |
| Printer | Thermal Transfer Overprinting (TTO) |